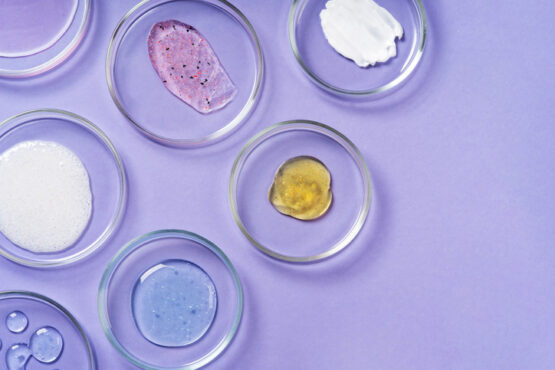
At the Edge of Innovation
Advanced Delivery Systems for Ambitious Projects in the USA
R&D + Manufacturing in the USA for oral care, sublingual films, multi-chamber systems, suppositories, topicals and dietary supplements

Who We Are
We are a US-based contract manufacturer that specializes in difficult, multi-component formulations — from professional in-office teeth-whitening systems engineered for summer heat-stability, to sublingual / orodispersible films (ODF) with high active loads, multi-chamber dispensers, suppository delivery systems, and dietary supplement innovations. We combine rapid prototyping, analytical rigor, and design-for-scale manufacturing to move ideas from lab to launch.
Why Customers Choose Us for the “Too Hard” Projects
Our Specialties: Oral-Care | Sublingual Film | Suppository | Dietary Supplement
If you’re expanding your product lines and need a partner who can formulate, scale, and manufacture reliably in the US, you’re in the right place. Atrium Scientific is best fit for: established brands and manufacturers who value Made in USA, cGMP quality systems, and repeatable output.
Quality, Compliance & Manufacturing
at US Scale
Facility: US-based, FDA-registered, cGMP compliant
QA/QC: Incoming, release testing, stability, retain program, CAPA
Packaging: Oxygen and moisture-barrier materials, dual-chambers, airless, pouches, stick packs, suppositories, capsules and unit-dose dispensers.
Supply chain: US and vetted global sources, alternate-vendor strategies to protect product launches.
Dietary Supplement Technologies
Atrium specializes in sublingual films, drinkable powders, and capsules, where bad-taste or ingredient stability would defeat regular formulations.

Fast-Dissolving ODF
High-loading up to 35% actives, taste-masking, high bioavailability.

Stick Packs
Label-friendly excipients, sugar-free systems, and stability systems.

Precise Capsules
Precise blending, consistent filling, strict quality control.
Suppository Delivery Systems
Lipid-base matrices, melting point tuning (33–37 °C), sedimentation control for suspended actives. Release profiling via in-vitro dissolution.
Unit-Dose
Leak-proof unit-dose packaging.

Automation
Fully-automated production lines.
Quality
Consistent & strict quality control.


How We Work
From Problem Statement to Launch at US Speed
Problem Framing (Week 0–1): Technical intake, target project summary, claims guardrails, and design for manufacturability (fill sizes, pack lines).
Rapid Prototyping (Weeks 1–6): Formulation sprints; build A/B variants internally around critical vectors – pH, rheology, stability, taste, performance panelist testing.
Analytical & Stress Testing (Weeks 4–8): Assay (titration / HPLC), rheometry (yield stress & thixotropy), accelerated stability (e.g., 50 °C), transport abuse (freeze-thaw), pH drifting, and packaging compatibility analysis (permeation, reaction, stability, etc.).
Scale-Up + IQ/OQ (Weeks 8–10): Pilot batches on coater/gel mixers, line trials on actual pouches/syringes/strips; set CQAs and in-process controls.
Launch (Weeks 10–16): Final specs, CoA, stability plan, artwork/label review; build safety & substantiation documentation.
Case Studies
Heat-Stable Professional Whitening Gel (No Cold Shipping Required)
A U.S. in-office whitening brand needed a high-potency peroxide gel that could withstand freight temperatures up to 50 °C without potency loss or phase separation. We engineered a water-based system using peroxide-compatible polymers and a radical-scavenger matrix to slow autocatalytic breakdown. The near-neutral pH improved patient comfort and minimized sensitivity. The result: <15% peroxide loss after 30 days at 50 °C, zero leakage, smooth non-drip chairside handling, and successful U.S. commercialization with repeat orders and a post-launch complaint rate below 0.3%.
High-Load Sublingual Film with Bitter Actives
A brand required a fast-dissolving oral thin film (ODF) delivering >35% active load while effectively masking strong bitter notes. We developed a sublingual film using taste-masking microencapsulation, an optimized PEG plasticizer system, and moisture-protective pouch packaging to preserve stability. The result: disintegration ≤40 seconds (USP <2040>), content uniformity CV ≤5%, and pouch residual moisture ≤3% at 6-month accelerated conditions. The product successfully scaled to continuous roll-to-roll manufacturing with >95% yield post die-cut.
Target Product Summary: claims, channel (professional/DTC/retail), pricing requirements
Functional requirements: actives, flavor, peroxide level (if any), film size / thickness
Performance targets: viscosity, pH, dosage, taste
Packaging preference: syringe/dual-chamber/strip/pouch/suppository
Timelines: prototype date, pilot, first PO
what to
bring
to your
scoping
call
Let’s Build Together
Based in the USA. Fast to sample. Built for scale.
We can start with a feasibility study or a full R&D program.
Email: info@atriumsci.com | Tel: (626) 380-5887